 
For example, in the water cycle, water repeatedly changes from a gas to a liquid or solid and back to a gas again. 
As matter moves through its cycles, it changes state repeatedly. Water and all the elements important to organisms, including carbon and nitrogen, are constantly recycled on Earth. Matter constantly goes through cycles that involve changing states (also called change phases). Technically speaking a fourth state of matter called plasma exists, but it does not naturally occur on earth, so we will omit it from our study here. Particles are free to move over each other, but are still attracted to each other Particles move in random motion with little or no attraction to each other No definite shape (takes the shape of its container) Table 1: Characteristics of each Phase of Matter Gases completely fill their containers, regardless of volume. Liquids have a fixed volume but flow to assume the shape of their containers. 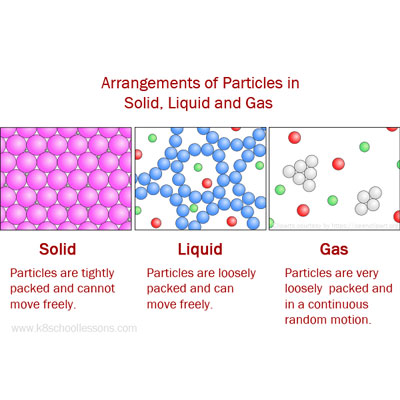
Increasing the pressure on a substance forces the molecules closer together, which increases the strength of intermolecular forces.įigure 2: The Three States of Matter. Heating and cooling can change the kinetic energy of the particles in a substance, and so, we can change the physical state of a substance by heating or cooling it. However, these also affect liquids and solids too. Gasses are very sensitive to temperatures and pressure. The intermolecular forces are attractive forces that try to draw the particles together (Figure 2). The kinetic energy keeps the molecules apart and moving around, and is a function of the temperature of the substance. The state of a substance depends on the balance between the kinetic energy of the individual particles (molecules or atoms) and the intermolecular forces. Images used with permission from Wikipedia. From the left, they are solid, liquid, and gas, represented by an ice sculpture, a drop of water, and the air around clouds, respectively. Figure 1: The three common states of matter. However, such changes of state do not affect the chemical composition of the substance. For example, liquid water can be heated to form a gas called steam, or steam can be cooled to form liquid water. Matter can often change from one physical state to another in a process called a physical change. Whereas the volume of gases strongly depends on their temperature and pressure (the amount of force exerted on a given area), the volumes of liquids and solids are virtually independent of temperature and pressure. Gases, such as air in an automobile tire, have neither fixed shapes nor fixed volumes and expand to completely fill their containers. In contrast, liquids have fixed volumes but flow to assume the shape of their containers, such as a beverage in a can. Solids are relatively rigid and have fixed shapes and volumes. Under normal conditions, there are three distinct states of matter: solids, liquids, and gases. For example, frozen nitrogen will form both the liquid phase and the vapor phase when exposed to normal temperature and pressure.\) Multiple phase changes can occur at once. For example, if you view the sublimation of dry ice into carbon dioxide gas, the white vapor that is observed is mostly water that is condensing from water vapor in the air into fog droplets. Phase changes aren't always clear when observing a situation. Plasma most often forms from ionization of a gas, although if sufficient energy and enough space are available, it's presumably possible for a liquid or solid to ionize directly into a gas. Plasma: Plasma can recombine to form a gas. Gases form from the sublimation of solids, vaporization of liquids, and recombination of plasma. Gases: Gases can ionize into plasma, condense into liquids, or undergo deposition into solids. Liquids form by condensation of gases and melting of solids. Liquids: Liquids can vaporize into gases or freeze into solids. Solids form by deposition from gases or freezing of liquids. Solids: Solids can melt into liquids or sublime into gases. Another way to list phase changes is by states of matter: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
